Troubleshooting blood smear errors
| Problem | Solutions |
|---|---|
| Short smear |
|
| Long smear/no feathered edge |
|
| Thick smear |
|
| Thin smear |
|
| Smear has waves and ridges |
|
Blood smears - common errors
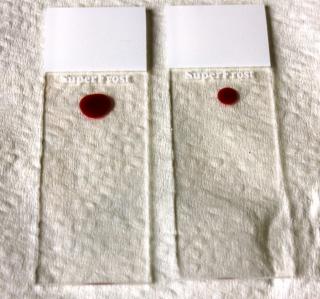
The blood droplet on the left is too big and will result in a thicker smear. The blood droplet on the right is the preferred size.
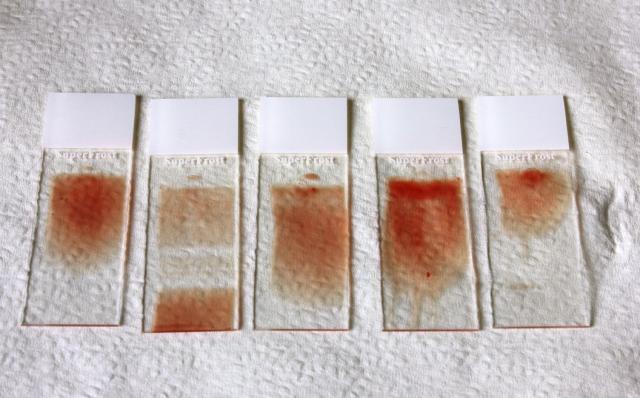
Smear technique - left to right:
- slide 1 - perfect smear
- slide 2 - smear technique interrupted in middle
- slide 3 - smear was skewed
- slide 4 - blood droplet too thick
- slide 5 - smear too short.
Infectious diseases in Australia diagnosed by blood smear
| Disease | Species | Transmission | Disease status |
|---|---|---|---|
| Anaplasmosis | cattle | tick-borne | endemic-northern Australia |
| Babesiosis | cattle | tick-borne | endemic-northern Australia |
| Mycoplasma ovis (eperythrozoonosis) | sheep | iatrogenic, blood-sucking insects (midges, mosquitoes, flies) | endemic |
| Bovine anaemia due to Theileria orientalis group (BATOG) | cattle | tick-borne | endemic in eastern Australia - first diagnosed in WA in 2013 |
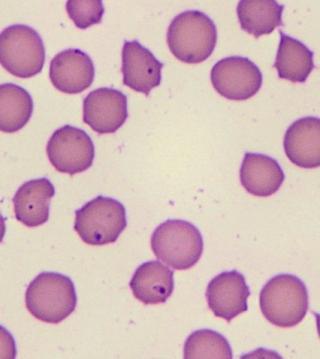
Bovine erythrocytes infected with protozoal parasites from the Theileria orientalis group. The parasite is known as a piroplasm when it is within an erythrocyte. Piroplasms appear in erythrocytes from day 10 post-infection. Naïve, young, pregnant or immunecompromised animals may develop severe anaemia and mortalities can be as high as 30% in a herd.
Ticks are vectors for many protozoal and mycoplasmal parasites. For Theileria spp., transmission in the tick is known to be transstadial or life stage-to-life stage. A larva or nymph stage tick transmits the parasite to the next animal it feeds on. Trans-ovarial transmission (transmission from infected females to their larvae) does not occur. Control of ticks and good sanitation when using needles and surgical equipment minimises the introduction of these infectious parasites into naïve populations.